Get Rid of Your Double Chin With This Nonsurgical Injectable Technique
What Is KYBELLA®?
KYBELLA® is a prescription medicine used in adults to improve the appearance and profile of moderate to severe fat below the chin (submental fat), also called “double chin.” It is not known if KYBELLA® is safe and effective for use outside of the submental area.

Who Should Not Receive KYBELLA®?
You should not receive KYBELLA® if you have an infection in the treatment area.
Before receiving KYBELLA®, tell us about all of your medical conditions, including if you: Have had or plan to have surgery on your face, neck, or chin, have had cosmetic treatments on your face, neck, or chin, have had or have medical conditions in or near the neck area, have had or have trouble swallowing, have bleeding problems, are pregnant or plan to become pregnant (it is not known if KYBELLA® will harm your unborn baby), are breastfeeding or plan to breastfeed (it is not known if KYBELLA® passes into your breast milk. Talk to us about the best way to feed your baby if you receive KYBELLA®).
Tell us about all the medicine you take, including prescription and over-the-counter medicine, vitamins, and herbal supplements. Especially tell us if you take medicine that prevents the clotting of your blood (antiplatelet or anticoagulant medicine).
What Are the Possible Side Effects of KYBELLA®?
KYBELLA® can cause serious side effects, including trouble swallowing and nerve injury in the jaw that can cause an uneven smile or facial muscle weakness.
The most common side effects of KYBELLA® include swelling, bruising, pain, numbness, redness, and areas of hardness in the treatment area.

Price:
$600/ Vial
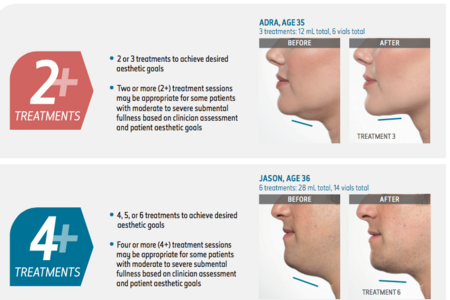
KYBELLA is injected into the subcutaneous fat tissue in the submental area using an area-adjusted dose of 2 mg/cm2.
A single treatment consists of up to a maximum of 50 injections, 0.2 mL each (up to a total of 10 mL), spaced 1 cm apart. (2 to 3 vials per session)
Up to 6 single treatments may be administered at intervals no less than 1 month apart.